Colorectal cancer (CRC) is the second leading cause of cancer death in the world. Standard treatment of patients with locally advanced cancer of the rectum includes chemoradiotherapy (CHRT) before surgery. In recent years, the new standard of treatment for some patients with CRC has become immunotherapy with the use of so-called immune checkpoint inhibitors (ICI) – drugs that ‘turn off’ cancer inhibition of the immune response. A particular group of patients who benefit from this treatment is characterized by the presence of tumours with impaired DNA repair mechanisms leading to the accumulation of mutations and the phenomenon of microsatellite instability. A consequence of these molecular features is a tumour more ‘foreign’ to the immune system, which is more infiltrated by lymphocytes and more susceptible to the effects of immunotherapy. Unfortunately, ICIs are not effective in treating patients whose tumours do not have these molecular characteristics.
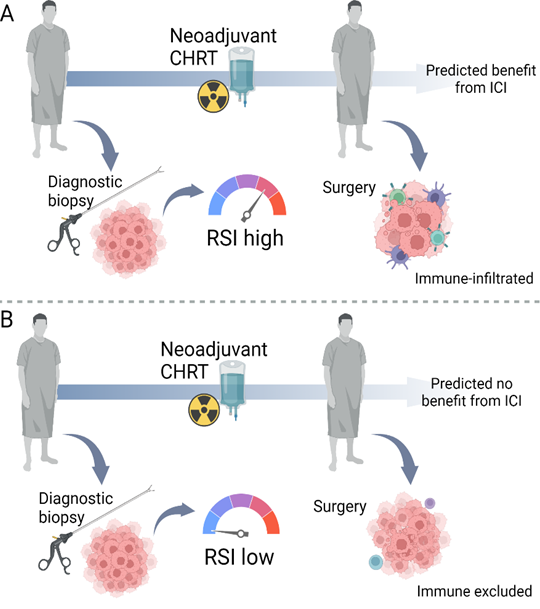
Recent studies have shown that chemotherapy and radiotherapy, treatments that are standard treatments for cancer, including rectal cancer, cause changes in the tumour environment that may partially mimic the nature of tumours that usually respond to immunotherapy. By killing cancer cells, they increase the level of compounds, including cytokines, that attract different populations of lymphocytes to the interior of the tumour, and also stimulate the presentation of antigens – i.e. molecular patterns that tell immune cells that cancer is foreign to the body. Based on these results, clinical trials began testing combinations of standard radio- and / or chemotherapy with immunotherapy. While the antitumor activity of these combinations has so far been demonstrated to be moderate, patients who successfully respond live notably long without the disease recurring. Therefore, it is very important, on the one hand, to design even better connections, and on the other hand, to search for markers – characteristics that indicate that a given patient and tumour will respond well to the combination of immuno- and radio / chemotherapy. One such marker may be the tumour’s intrinsic radiosensitivity index (RSI), i.e. molecularly determined susceptibility to radiation.
The proposed study will check whether rectal tumours with a higher RSI level examined at diagnosis respond to preoperative chemoradiotherapy in a way that suggests that immunotherapy after or in combination with conventional treatment may be effective for these patients. We will also carefully examine various characteristics of tumours, including the level of proteins responsible for antigen presentation and infiltration by different lymphocyte populations, in paired samples before and after chemoradiotherapy. This will allow for the design of better combinations with immunotherapy in the future and a more optimal choice of patients for whom new treatment regimens should be tested.
This project is funded by National Science Centre (NCN) grant Preludium 21 no. 2022/45/N/NZ5/01240, for more information visit here.